 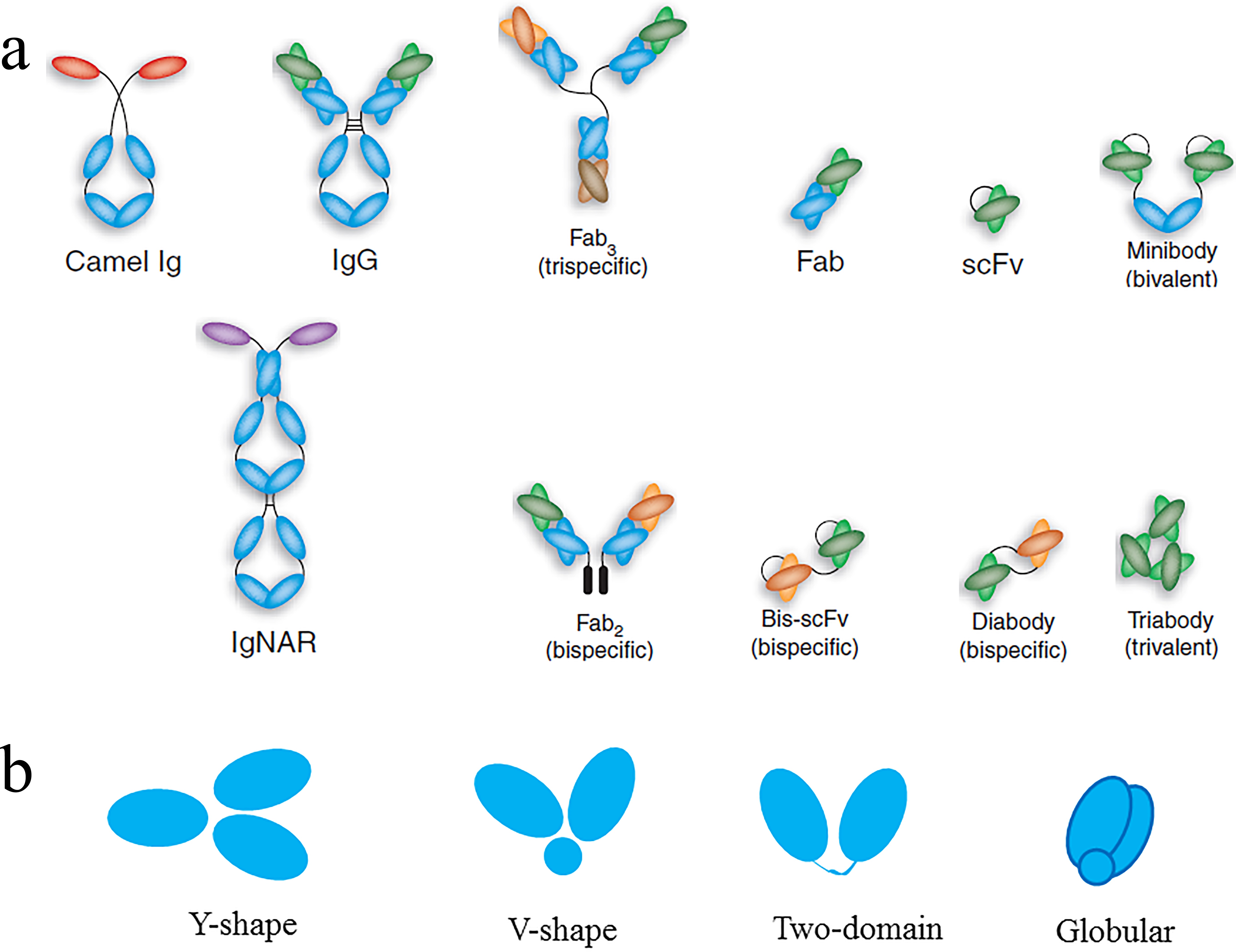 
IgAs, IgDs, and IgGs have three constant (C) and one variable (V) domains. In comparison, human antibody HCs can be one of five isotypes, IgA, IgD, IgE, IgG, and IgM, each with an independent role in the adaptive immune system. Both LC classes have two domains, a constant domain (CL) and a variable domain (VL). Human LCs can be one of two functionally similar classes, κ or λ. The two HCs of the heterotetramer are also linked by disulfide bridges. The HC and LC of the heterodimer are linked through disulfide bonds. In natural systems, the pairing of one LC with one HC associates with another identical heterodimer to form the intact immunoglobulin. Human immunoglobulins are Y-shaped proteins composed of two identical light chains (LCs) and two identical heavy chains (HCs). The overall structure of antibodies, including the folding pattern of the individual domains and basic features of the antigen-combining sites, has been the subject of several reviews. The structural data includes complexes of these molecules with proteins, other macromolecules, peptides, and haptens. At present, the Protein Data Bank (PDB) contains over 3500 structures of antibody fragments (Fabs, Fvs, scFvs, and Fcs), as well as a small number of intact antibody structures. Our knowledge of the three-dimensional structure of antibodies has emerged from crystallographic studies reported from numerous laboratories beginning in the 1970s. The engineering approaches being used are based on our knowledge of protein structure and, in particular, our knowledge of how the structures are linked to their function. Different strategies of preparing bispecific and multispecific molecules for an array of therapeutic applications are included.Ĭurrently, all antibodies and antibody-derived macromolecules being developed for a wide spectrum of therapeutic indications require protein engineering. We also review the design and selection of binding arms, and avidity modulation. The platforms examined include the development of antibodies, antibody fragments, bispecific antibody, and antibody fusion products, whose efficacy and manufacturability can be improved via humanization, affinity modulation, and stability enhancement. In this review, our basic understanding of the antibody structure is described along with how that knowledge has leveraged the engineering of antibody and antibody-related therapeutics having the appropriate antigen affinity, effector function, and biophysical properties. Our knowledge of the structure–function relationships of antibodies provides a platform for protein engineering that has been exploited to generate a wide range of biologics for a host of therapeutic indications. Immunol.Antibodies and antibody-derived macromolecules have established themselves as the mainstay in protein-based therapeutic molecules (biologics). Tonegawa S (1993) Somatic generation of immune diversity. 
Tonegawa S (1983) Somatic generation of antibody diversity. Sanger F, Nicklen S, Coulson AR (1977) DNA sequencing with chain-terminating inhibitors. Radic MZ, Zoulali M (1996) Receptor editing, immune diversification, ana self tolerance. Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning- a laboratory manual. Gubbler U, Hoffman BJ (1983) A simple and very efficient method for generating cDNA libraries. (1989) Conformations of immunoglobulin hypervariable regions. 105: 5–26 G-Ĭhothia C, Lesk AM, Tramontano A, Levitt M, Smitn-Giiair G, Sneriff S, Raulan EA, Davies D, Tulip WR, et al. This process is experimental and the keywords may be updated as the learning algorithm improves.īerek C, Milstein C (1988) The dynamic nature of the antibody repertoire. These keywords were added by machine and not by the authors. The diversity in this site enables antibodies to recognise a wealth of antigenic structures. The antigen binding site is formed by the association of six polypeptide loops, three from the variable domain of the heavy chain (H1, H2 and H3) and three from the variable domain of the light chain (L1, L2 and 元) (see Chothia et al., 1989). The immunoglobulin molecule is bifunctional: the variable domains are involved in binding to the antigen, whereas the constant domains mediate effector functions (interaction of the antibody with other components of the immune system, such as phagocytic cells and/or complement). 
Each heavy chain consists of a variable domain (V H) and three or four constant domains (C H) each light chain consists of a variable domain (V L: V K or V λ) and a single constant domain (C L: C K or Cλ). The antibody molecule, or immunoglobulin (Ig), is composed of two identical heavy (H) and two identical light (L) polypeptide chains, with the light chains being of either kappa (k) or lambda (λ) type. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
